Publications
When it comes to fighting for health freedom, information is key and knowledge is power. You can find CCHF’s latest content – as well as our archived content – here on our website. From original papers and articles to issues of our quarterly Health Freedom Watch, this is where we add published content to keep you informed.

Key Initiatives
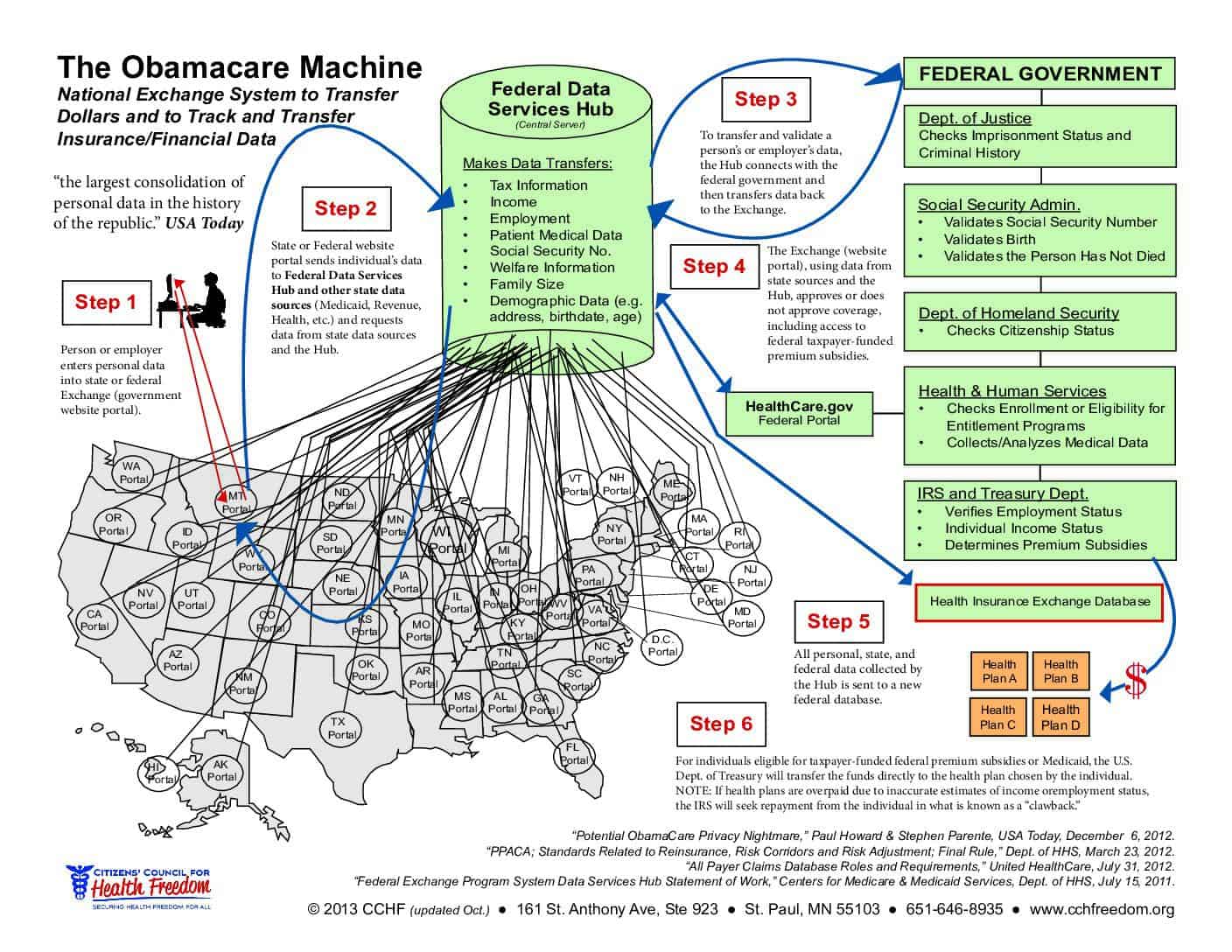
HIPAA: The Deliberate Deception
HIPAA is a permissive data-sharing rule that does not protect your privacy.

REAL ID - Don't Comply
Unconstitutional National ID card paving the way to a possible China-like Digital Social Credit System of control.

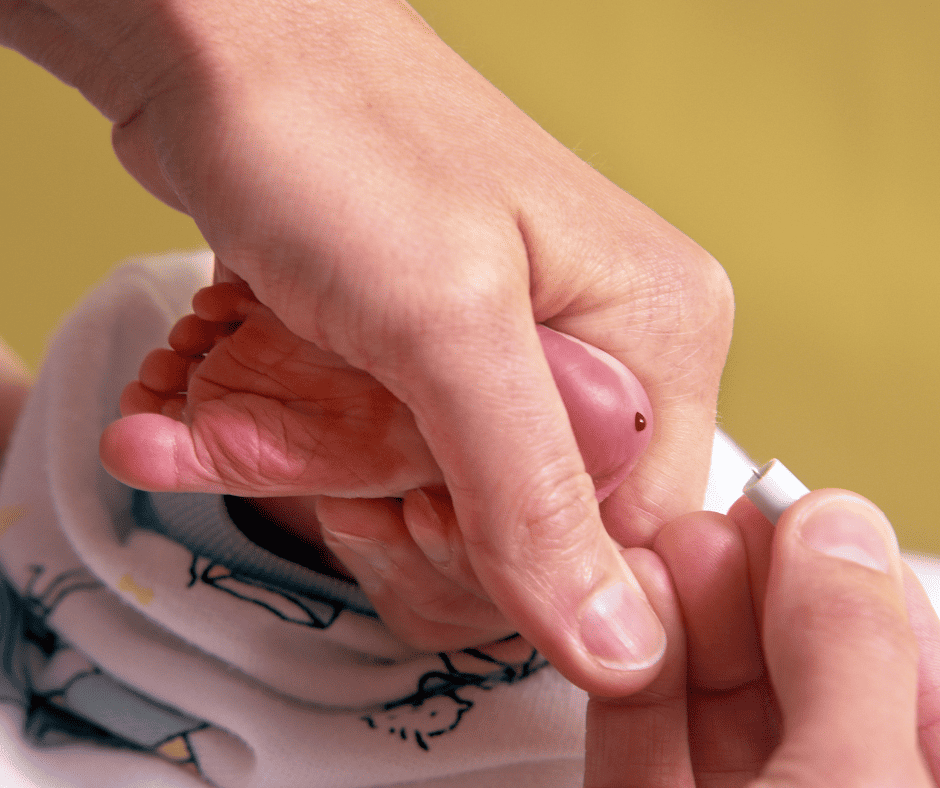
Taking Baby DNA
Government storage of newborn DNA without parental consent after
newborn screening.

The Wedge of Health Freedom®
See our online network of cash-based practices – find a doctor who works for you!

Medicare Opt-Out
Giving seniors the right to opt out of Medicare without losing access to Social Security Retirement Benefits

Exam Room Access for Parents
COMING SOON:
Giving parents the right to stay in the exam room with their teenage children
Subscribe to the Newsletter
Enter your email to begin receiving monthly newsletter emails.